|
Sometimes environmental factors, especially low pH and high temperatures, alter the shape of the active site if the shape cannot be restored, the enzyme denatures. For example, trypsin, an enzyme in the human small intestine, works most efficiently at pH 8, whereas pepsin in the stomach works best under acidic conditions. Enzymes work most efficiently under optimal conditions that are specific to the enzyme. An allosteric site is an alternate part of the enzyme that can bind to non–substrate molecules. 
Inhibitors, coenzymes, and cofactors can act competitively by binding to the enzyme’s active site, or noncompetitively by binding to the enzyme’s allosteric site. After the reaction finishes, the product(s) are released, and the active site returns to its original shape.Įnzyme activity, and thus the rate of an enzyme-catalyzed reaction, is regulated by environmental conditions, including the amount of substrate, temperature, pH, and the presence of coenzymes, cofactors, activators, and inhibitors. Substrate binding alters the shape of the enzyme to facilitate the chemical reaction in several different ways, including bringing substrates together in an optimal orientation. One part of the enzyme, the active site, interacts with the substrate via the induced fit model of interaction. Enzymes are very specific for the reactions they catalyze because they are polypeptides, enzymes can have a variety of shapes attributed to interactions among amino acid R-groups. 
Enzymes are macromolecules-most often proteins-that speed up chemical reactions by lowering activation energy barriers. Sucrase is an example of an enzyme, a type of biological catalyst. 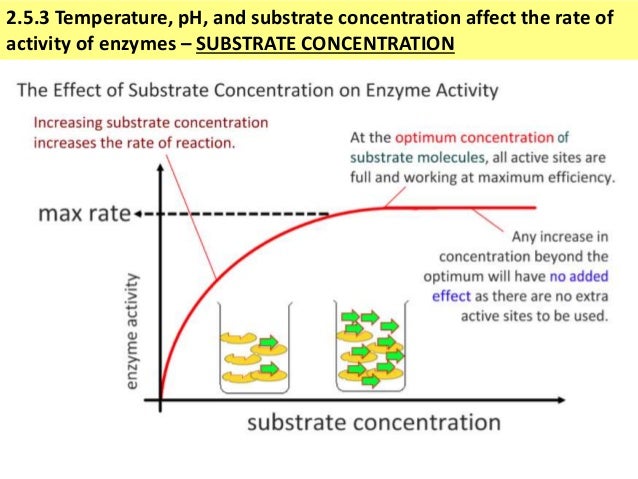
For example, a teaspoon of sucrose (table sugar), a disaccharide, in a glass of iced tea will take time to break down into two monosaccharides, glucose and fructose however, if you add a small amount of the enzyme sucrase to the tea, sucrose breaks down almost immediately. Many chemical reactions in cells occur spontaneously, but happen too slowly to meet the needs of a cell.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
